Schweitzer P.A. Fundamentals of corrosion. Mechanisms, causes, and preventative methods
Подождите немного. Документ загружается.


Corrosion of Paint 229
7.6.3.5 Other Treatments
Procedures have been developed specically for plastic processing to over-
come the low surface activity of many of these materials. Prior removal of
surface contamination by solvent or detergent cleaning is necessary in most
cases to achieve optimum results.
Primers.• The application of a primer coating is used to develop bet-
ter adhesion of the nal coating to the plastic substrate. A variety
of chemical types can be used as primers, including urethane poly-
mers, silicones, nitriles, phenolics, vinyls, or isocyanates. The primer
is applied to the surface as soon as possible after other surface prepa-
ration procedures have been completed in order to protect the sur-
face from recontamination.
Flame treatment.• The surface of many plastics, such as acetals, poly-
olens, uoropolymers, and polycarbonates, are oxidized by the
impingement of a ame. The oxidation provides a higher level of
surface energy and better lm adhesion. This process is particularly
effective on complex shapes and molded parts. Superheated air at
1000°F (538°C) is just as effective.
Exposure to UV radiation.• An ionized or highly polar surface results
after exposure to high-intensity UV radiation.
Drying.• Over-drying can be effective on plastic formulations that
absorb atmospheric moisture.
Plasma treatment.• Plasma treatments are effective for most plastics.
Parts are exposed to gases such as neon, helium, oxygen, and mois-
ture vapor, which are ionized by radio-frequency or microwave
discharge. Although very effective in improving surface conditions
that promote better lm adhesion, this process is limited to smaller
components and parts because of equipment size.
Corona discharge.• Surface tension can be improved by passing lm or
thin-gauge plastics between two electrodes. This treatment is suit-
able for high-speed operations.
7.7 Application of Coatings
Over the years there have been many changes in the formulation of coat-
ings that have affected the methods by which they are applied to a substrate.
Several of these changes have resulted from governmental regulations.
The Occupational Safety and Health Administration (OSHA) and the Toxic
Substances Control Act (TSCA) regulate the environment in the workplace
230 Fundamentals of Corrosion
and limit workers’ contact with hazardous materials. OSHA and the TSCA
make it necessary to use alternate coating materials and to modify applica-
tion procedures in order to comply.
In the late 1940s, smoke control laws were enacted to reduce airborne
particulates that led to air pollution. During this period, a condition
known as “photochemical fog” developed as a result of increased automo-
bile usage and industrial expansion. The fog was created by the reaction of
chemicals in the atmosphere to sunlight. Los Angeles County ofcials, rec-
ognizing that automobile exhaust and VOC (volatile organic compound)
emissions were major sources of smog, enacted an air pollution regulation
called Rule 66. Under Rule 66, specic solvents that produce photochemi-
cal smog were banned from use. At the same time, they published a list
of acceptable solvents that could be used in coatings. The EPA conducted
additional studies that indicated that these so-called “acceptable” sol-
vents, given enough time, would also produce photochemical smog in the
atmosphere.
The EPA established national air quality standards (in its Clean Air Act of
1970 and its 1990 amendments) that regulate the amounts of solvents that can
be emitted. Many local standards are more stringent than the national stan-
dard. Consequently, specic coatings may not comply with regulations in all
areas. Waterborne, powder, high-solids, electropheric, and radiation-cured
coatings will comply in all areas.
In addition, certain types of paints, primarily those containing lead and
asbestos, have been outlawed by federal and local jurisdictions. Potentially
harmful pigments or other constituents are causing the restriction of other
paints containing these ingredients.
It is important that, during the application of any coating, adequate ven-
tilation for the removal of solvents be provided, as well as suitable and safe
access to the work being painted.
7.7.1 application Methods
The method of application of a corrosion-resistant paint will depend on the:
1. Purpose for which coated product will be used
2. Environment to which the coating will be exposed
3. Type of paint
4. Shape and size of the object to be painted
5. Period of application process
6. Cost
Corrosion of Paint 231
Application methods vary from the traditional paint brush or roller to vari-
ous spray techniques, powder coating, and electrodeposition. Exact proce-
dures will depend on the specic coating used and the object to be coated.
7.7.1.1 Brushing
Brushing is an effective, relatively simple method of paint application, par-
ticularly with primers, because of the ability to work the paint into pores
and surface irregularities. Because brushing is slow, it is used primarily for
smaller jobs, surfaces with complex congurations (edges, corners, cuts, etc.),
or where overspray might cause a serious problem.
Brushing was once the main coating method but, at the present, spray coat-
ing is more widely used. Brush coating has the following advantages:
1. Applicators are simple and inexpensive.
2. Complicated forms and shapes can be coated.
3. Thick lms are obtained with one coat.
4. Particularly useful in applying an anti-rust coating.
The disadvantage of brushing results from the nonuniformity of coating lay-
ers, especially coating layers of rapidly drying paints.
7.7.1.2 Rolling
The advantage of using rollers is found when used on large, at areas that
do not require the smoothness or uniformity that can be obtained by spray-
ing. They are also used in interior areas, where overspray presents a clean-
ing and masking problem. Because of the difculty of penetrating pores,
cracks, and other surface irregularities, the use of a brush is preferred when
applying primers. When using a roller, air mixes with the paint and leaves
points where moisture can penetrate the cured lm. Rolling is best when
used to apply a topcoat over a primer that has been applied by some other
method.
7.7.1.3 Roller Coating
Roller coating is a process to coat coils and sheets by passing them through
two preset rollers. The quantity of paint consumed is reduced by approxi-
mately 50% of that used with other coating methods. The thickness of the
lm is controlled by adjusting the rollers. One-side or both-side coatings are
possible.
232 Fundamentals of Corrosion
7.7.1.4 Spray Painting
A more uniform and smoother surface can be achieved with spray painting
than with brushing or rolling because the latter methods tend to leave brush
or stipple marks with irregular thickness. The most common methods of
spray painting are conventional and airless.
The conventional spray method relies on air for paint atomization. Jets
of compressed air, introduced into the stream of paint at the nozzle, break
the stream into tiny droplets that are carried to the surface by the air cur-
rent. Paint losses from bounce-back or overspray can be high because large
amounts of air are mixed with the paint during application. Such losses have
been estimated to be as much as 30 to 50%.
Some of the disadvantages of conventional air spray applications include:
1. It is slower than airless application.
2. More overspray results than from other methods.
3. It is difcult to coat corners, crevices, etc., because of blowback.
An airless spray system consists of:
1. A plunger pump that supplies high pressure to the paint
2. An airless spraygun
3. A high-pressure-resistant hose
High-viscosity paints are warmed before spraying. This technique has the
following advantages over an air spray system:
1. The sticking rate of paint is increased by 24 to 40%.
2. A thicker lm can be applied.
3. The running of paint on the substrate is reduced.
4. Because there is only one hose, it is easier for the operator to use.
5. Higher viscosity paints can be applied.
6. Clean-up is easier.
Because the airless spray process is more efcient than brushing, it is suit-
able for coating steel structures and bridge girders in the factory. However,
paint loss using the airless spray process is 30 to 40% greater than that of
brushing. The disadvantages of the airless spray system include:
1. Reliance on dangerous high pressure
2. Fan pattern is not adjustable
3. Additional working parts that can cause difculty
Corrosion of Paint 233
4. Higher initial cost than other spraying techniques
5. A need to exercise extra care to avoid excessive buildup of paint that
causes solvent entrapment, pinholes, runs, sags, and wrinkles
7.7.1.5 Powder Coating
Powder coatings have grown in popularity as anti-pollution coatings
because of the absence of solvents. Coating thicknesses of 25 to 250 μm can
be obtained.
Automotive bodies, electric components, and housing materials, wire and
cables make use of this process. Polyethylene and epoxy resins are the pre-
dominant types of paints used. At the present time, the following proce-
dures are used:
1. Pouring method (ock coating)
2. Rotational coating of pipes
3. Fluidized bed
4. Dipping in nonuidizing powders
5. Centrifugal casting
6. Rotational molding
7. Electrostatic powder spraying
8. Electrostatic uidized bed
9. Pouring or owing of uidized powder
10. Electrogas dynamics powder spraying
11. Flame spraying of thermoplastic powders
Powder coating was developed in the 1950s and is a method for applying
nely divided, dry, solid, resinous coatings by dipping products in a uidized
bed or by spraying them electrostatically. The uidized bed is essentially a
modied dip tank. During the electrostatic spraying method, charged par-
ticles adhere to grounded parts until fused and cured. In all cases, the pow-
der coating is heated to its melt temperature, where a phase change occurs,
causing it to adhere to the product and fuse to form a continuous coating.
The uidized bed powder coating is a dipping process using dry, nely
divided plastic materials; a tank having a porous bottom plate forms the
uidized bed. The plenum below the porous plate uniformly supplies low-
pressure air across the plate, which suspends the nely divided plastic pow-
der particles. Products that are preheated above the melt temperature of the
material are dipped into the bed where the powder melts and fuses into a
continuous coating. Thermosetting powders often require additional heat to
cure the lm on the product.
234 Fundamentals of Corrosion
Fluidized bed powder coating has the advantage of producing a uniform
and reproducible lm thickness. It also has the advantage of producing a
heavy coating in one dip. The disadvantage of this method is the 3-mil mini-
mum lm thickness required to form a continuous coating.
An electrostatic uidized-bed coater is essentially a uidized bed with a
high-voltage DC grid installed above the porous plate to charge the nely
divided particles. The charged particles repel the grid and each other, form-
ing a cloud. These particles are attracted to and coat products that are at
ground potential. Film thicknesses of 1.5 to 5 mil are possible on cold parts
and 20 to 25 mil are possible on heated parts.
The advantage of this method is that small parts such as electrical com-
ponents can be coated uniformly and quickly. The disadvantage is that the
product size is limited and inside corners have low lm thickness.
Electrostatic spray coating is a method for applying nely divided, elec-
trostatically charged particles to products that are at ground potential. A
powder/air mixture from a small uidized bed in a powder reservoir is
supplied by hose to a spray gun that has a charged electrode in the nozzle
fed by a high-voltage DC power pack. The spray guns can be manual or
automatic and mounted in a conveyorized spray booth. Film thicknesses
of 1.5 to 5 mil can be obtained on cold substrates. A 20- to 25-mil thickness
can be obtained on heated substrates. The advantage of this method is that
coatings using many resin types can be achieved in thicknesses of 1.5 to 3
mil, with no VOC emissions. Disadvantages include the difculty in obtain-
ing a continuous coating of less than 1 mil; and because of complex powder
reclaiming systems, color changes are more difcult to make than with liq-
uid spray systems.
7.7.1.6 Electrodeposition of Polymers
The electrodeposition of polymers is an extension of painting techniques
into the eld of plating and, like plating, is a dip coating process. In the case
of ionizable polymers, the deposition reaction is:
RNHOHIFRNHO
33
+−
+→ +
2
or the conversion of water-dispersed ammonium-type ions into ammonia-
type, water-insoluble polymers known as cathodic deposition. Alternatively,
a large number of installations utilize the anodic deposition process:
RCOOH HlessIFRCOOH
+
+→
Corrosion of Paint 235
where R symbolizes any of the widely used polymers (such as acrylics, epox-
ies, alkyds, etc.). The electrodeposition process is dened as the utilization of
synthetic, water-dispersed, electrodepositable macroions.
Metal ions, typically 0.5Ni
2+
, show an electrical equivalent weight equal to
approximately 29.5 g while the polymeric ions typically used for electrode-
position exhibit a gram equivalent weight (GEW) of approximately 1600.
Therefore, IF plates out 30 g nickel and deposits 1600 g macroions.
The formation of uniformly thick coats on all surfaces of a formed work
piece, including the extreme recesses, such as the inside of car doors, is prob-
ably the reason for the rapid industrial growth of this process. The ability to
extend coats into recesses is known as “throwing power.”
Another advantage is the very small emission of volatile organic com-
pounds (VOCs), making electrodeposition with powder coating and radia-
tion cure the least polluting coating process.
The anodic deposition process for paint coating systems was introduced in
the early 1960s, and the cathodic deposition process in 1972. Electrodeposition
processes are widely used because they possess the advantages of unmanned
coating, automation, energy savings, and lower environmental pollution.
This process is used to apply coatings to automotive bodies and parts, domes-
tic electrical components, machine parts, and architecturals such as window
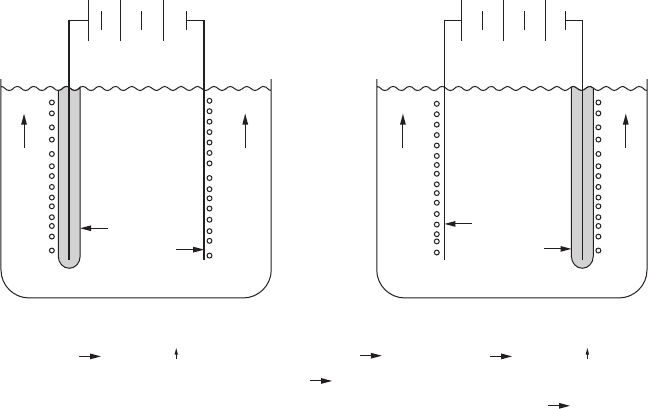
frames. Schematic illustrations of anodic and cathodic electrodeposition of
paints are shown in Figure 7.4.
The primary paints used in the electrodeposition process are anionic-type
resins with a carboxyl group (RCOOH polybutadiene resin) and cationic-type
resins (R-NH
2
epoxy resin). Hydrophilic groups and neutralizing agents are
added to the water-insoluble or undispersed prepolymers to convert them to
soluble or dispersed materials.
The dissolution of metal substrate in the cathodic process is much less than
that in the anodic process. The primary resins used in the cathodic process are
epoxy; and because epoxy resins provide good water and alkali resistance as
well as adhesion, cationic paint coatings are superior to anodic paint coatings.
7.7.2 Multilayer Coatings
The thicker a coating, the better the protective ability. However, the thickness
of a single coat is restricted because thick paint lms tend to crack as a result
of the internal stress. When a product is to be used for an extended period
of time in a severe environment, multilayer paint coating systems are usu-
ally employed. Automotive bodies and steel structures are typical products
receiving multilayer coatings. A two- or three-coat system is employed for
automotive bodies, and a general or heavy-duty coating system is adopted
for steel structures. A typical paint coating system for an automotive body
is as follows:
236 Fundamentals of Corrosion
1. Pretreatment (degreasing and phosphating) by dipping or spraying
2. Primer coating by cathodic electrodeposition
3. Sealing by blow gun or undercoating by air spray
4. Surface conditioning by atting
5. Intermediate spray coating by auto-electrostatic powder process or
spraying
6. Surface conditioning by atting and wax injection
7. Top paint coating by auto-electrostatic powder process, as by spray
The paint system for auto bodies is composed of a combination of various
types of paint and effective coating processes, thereby providing optimal
corrosion protection and decorative appearance.
The paint system for a steel structure is selected based on the required ser-
vice life and environmental conditions under which the steel structure must
exist. Structures in mild environments are commonly coated with general
coating systems. Those in severe environments are treated with heavy-duty
coating systems. Typical paint coating systems for steel structures are given
in Table 7.11.
Anodic reactions
Deposition of film
2H
2
O4H
+
O
2
4e
–
+
+
Anodic reactions
Deposition of film
2H
2
O4H
+
O
2
4e
–
+
+
Cathodic reactions
Cathodic reaction
2H
2
O 4OH
–
2H
+
H
2
2e
–
O
2
4e
–
+
+
+
2H
2
H
2
2e
–
+
(–)
(+)
(–)
(+)
O
2
H
2
OH
–
Deposition
of coating
film
Deposition
of coating
film
Anodic Cathodic
H
+
H
2
OH
2
O
O
2
H
2
OH
–
H
+
(a) (b)
FigurE 7.4
(a) Anodic and (b) cathodic electrodeposition of paints.

Corrosion of Paint 237
TabLE 7.11
Application Examples of Multistage Paint Coating Systems for Steel Structures
General Coating System
A B C
1st coat Etching primer Etching primer Zinc-rich primer
2nd coat Oil corrosion
preventive paint
Oil corrosion
preventive paint
Chlorinated
rubber system
primer
3rd coat Oil corrosion
preventive paint
Oil corrosion
preventive paint
Chlorinated
rubber system
primer
4th coat Long-oil alkyd
resin paint
Phenolic resin
system M10
paint
Chlorinated
rubber system
paint
5th coat Long-oil alkyd
resin paint
Chlorinated
rubber system
paint
Chlorinated
system rubber
paint
6th coat Chlorinated
rubber system
paint
Heavy Coating System
D E F G
1st coat Zinc spray or
zinc-rich paint
Thick-type
zinc-rich paint
Thick-type
zinc-rich paint
Zinc-rich primer
2nd coat Etching primer Thick-type vinyl
or chlorinated
rubber system
paint
Thick-type epoxy
primer
Tar epoxy resin
paint
3rd coat Zinc chromate
primer
Thick-type vinyl
or chlorinated
system rubber
paint
Thick-type epoxy
primer
Tar epoxy resin
paint
4th coat Phenolic resin
system M10
paint
Vinyl or
chlorinated
rubber system
paint
Epoxy resin
system paint
Tar epoxy paint
5th coat Chlorinated
rubber system
paint
Vinyl or
chlorinated
rubber system
paint
Epoxy or
polyurethane
resin system
paint
6th coat Chlorinated
rubber system
paint

238 Fundamentals of Corrosion
7.7.3 Curing
For a coating to be effective, it must be properly cured. Unless this is allowed
to take place, the coating will not provide the protection required nor have
the expected lifetime.
Most organic resins are liquid, which cure or dry to form solid lms. They
are classied as thermoplastic or thermosetting. Thermoplastic resins dry by
solvent evaporation and will soften when heated and harden when cooled.
Thermosetting resins will not soften when heated after they are cured. Most
organic resins are affected by solvents. Table 7.12 lists organic resins and
solvents that affect them.
Coatings are also classied by their various lm-forming mechanisms,
such as solvent evaporation, coalescing, phase change, and conversion.
Additionally, they are classied as room-temperature curing (sometimes
called air-drying) or heat curing (generally referred to as baking or forced dry-
ing), which uses elevated temperatures to accelerate drying. Thermoplastic
and thermosetting coatings can be both air-drying and baking.
TabLE 7.12
Solvents That Affect Organic Resins
Resin
Heat-Distortion
Point (°F) Solvents that Affect Surface
Acetal 338 None
Methyl methacrylate 169–195 Ketones, esters, aromatics
Modied acrylic 170–190 Ketones, esters, aromatics
Cellulose acetate 110–209 Ketones, some esters
Cellulose propionate 110–250 Ketones, esters, aromatics,
alcohols
Cellulose acetate butyrate 115–227 Ketones, esters, aromatics,
alcohols
Nylon 260–360 None
Polyethylene:
High density 140–180 None
Medium density 120–150 None
Low density 105–121 None
Polypropylene 210–230 None
Polycarbonate 210–290 Ketones, esters, aromatics
Polystyrene (G.P. high heat) 150–195 Some aliphatics, ketones,
esters, aromatics
Polystyrene (impact, heat
resistant)
148–200 Ketones, esters, aromatics,
some aliphatics
ABS 165–225 Ketones, esters, aromatics,
alcohol
