Schweitzer P.A. Fundamentals of corrosion. Mechanisms, causes, and preventative methods
Подождите немного. Документ загружается.

Atmospheric Corrosion 109
protective lm on the base metal. Due to the lack of protective lm forma-
tion, white rust corrosion will continue until the protective zinc coating is
entirely removed from the underlying steel, which is then subjected to accel-
erated corrosion and premature failure. White rust will occur whenever zinc
or galvanized steel is exposed to water that has a pH value above 8.2. The
corrosion rate is governed by the alkalinity of the water and the presence of
any accelerating agents. Why the basic zinc carbonate cannot form has not
been established. It may be due to long-lasting moistening or to a poor sup-
ply of CO
2
. White rust is more prevalent during rainy seasons. It may also
form on moist zinc surfaces in contact with mineral wool, which prevents
access of air and drying. This condition may happen in heat-insulated wall
structures containing galvanized steel members.
In years gone by, zinc coating of steel (galvanizing) was considered a sat-
isfactory corrosion-resistant material for structures in outdoor atmospheres.
The corrosion rate was only 0.5 to 1.0 µm/yr. However, the corrosion rate has
been increasing in many urban or industrial areas as a result of increasing
pollution by SO
2
. Corrosion rates have reached 5 µm/yr or more in many
areas. Because of this, it is often necessary to apply to galvanized steel a coat
of anticorrosion paint for added protection.
4.7.4 Zinc and Zinc alloy Coatings on Steel
Galvanized steel is steel that has been coated with zinc. The galvanizing pro-
cess is widely used to protect steel from atmospheric corrosion. Structures,
sheet steel, wire, and piping are all forms that are protected by galvanizing.
The protection afforded in rural areas is greater than that in urban or
industrial atmospheres. In the latter areas there is a greater concentration
of industrial pollutants. The air in these areas is contaminated with various
sulfur compounds, which together with the moisture in the air convert the
normally impervious corrosion-resistant zinc carbonate and zinc oxide layer
into zinc sulfate and zinc sulte. These water-soluble compounds have poor
adhesion to the zinc surface and therefore are washed away relatively easily
by rain. This exposes the underlying surface to attack by oxygen in the air.
A galvanized steel coating normally corrodes in the same manner as solid
zinc. In areas where the zinc coating is defective, the exposed steel, under
most conditions, is cathodically protected. Because of the iron-zinc alloy
present in most galvanized coatings, galvanized surfaces have a higher cor-
rosion resistance than zinc in neutral and acid conditions. Zinc coatings on
steel exposed to air corrode in the same manner as solid zinc, and there is an
approximate linear relationship between weight loss and time. In industrial
atmospheres, the purity of the coating has little effect on the corrosion rates.
Protection of galvanized surfaces from atmospheric corrosion is due to the
formation of basic salts, primarily carbonate. The most widely accepted for-
mula is Zn
5
(OH)
6
(CO
3
)
2
. The galvanized surface of zinc is rapidly attacked
when environmental conditions lead to the formation of soluble lm. One of
110 Fundamentals of Corrosion
the most important factors affecting the corrosion of zinc in the atmosphere
is the duration and frequency of moisture contact.
Because a thin lm of moisture with a high oxygen concentration promotes
corrosion, the rate of drying time is an important factor. Under normal expo-
sure conditions, the lms dry rapidly, and it is only in sheltered areas that
drying times are so slow that the attack of zinc is accelerated.
The effect of atmospheric humidity on the corrosion of zinc is related to the
conditions that may cause condensation of moisture on the metal surface and
to the frequency and duration of the moisture contact. Moisture is deposited
when the air temperature drops below the dew point. The quantity of dew
deposited is inuenced by the thickness of zinc, and its surface roughness
and cleanliness. When the temperature of the metal surface falls below the
air temperature in a humid atmosphere, moisture condenses on the metal.
If the water evaporates quickly, corrosion is not severe and a protective lm
forms on the surface. If water from rain or snow remains in contact with zinc,
when access to air is restricted and the humidity is high, the resulting corro-
sion can appear to be severe because the formation of a protective basic zinc
carbonate is prevented.
Atmospheric corrosion of zinc is roughly proportional to the time of wet-
ness in a particular location, provided the nature and quantity of environ-
mental pollution do not change. A high relative humidity, which can cause
condensation, increases corrosion.
Time of wetness is normally secondary to the effect of atmospheric pol-
lutants, primarily sulfur dioxides and other acid forming pollutants, which
have a direct effect and can also make rain more acidic. In less corrosive
areas, time of wetness assumes a greater proportional signicance.
Sulfur dioxide is a primary pollutant leading to the atmospheric corrosion
of zinc. It controls the corrosion rate when the relative humidity is in the
area of 70% or above. Sulfur oxides and other air pollutants are deposited on
zinc surfaces either by dry or wet deposition. Regardless of the method of
deposition, the sulfur dioxide deposited on the zinc surface forms sulfurous
or other strong acids, which react with the protective zinc oxide, hydroxide,
or basic carbonate lm to form zinc sulfate. The lm of protective corro-
sion products is destroyed by the acids, which reforms from the underlying
metal, causing the continuous corrosion by an amount equivalent to the lm
dissolved, and hence to the amount of sulfur dioxide absorbed. Corrosion
rates increase even further when the relative humidity exceeds 85%.
Atmospheric chlorides will lead to the corrosion of zinc, but to a lesser
degree than the corrosion of steel, except in brackish water and owing sea-
water. Any salt deposit should be removed by washing. Chlorides also have
a much smaller effect on the corrosion rate of zinc than sulfur compounds.
The salt content of the atmosphere will usually decrease rapidly inland fur-
ther away from the coast. Corrosion also decreases with distance from the
coast but the change is more gradual and erratic because chloride is not the
primary pollutant affecting zinc corrosion. Chloride is most harmful when
Atmospheric Corrosion 111
combined with acidity resulting from sulfur gases, as would be found in
highly industrialized coastal areas.
Other pollutants also have an effect on the corrosion of galvanized sur-
faces. Deposits of soot or dust can be detrimental, both because of their
specic nature and because they have the potential to increase the risk of
condensation on the surface and hold more water in position. This is preva-
lent on upward-facing surfaces. Soot (carbon) absorbs large quantities of sul-
fur, which are released by rainwater.
In rural areas, over-manuring of agricultural land tends to increase the ammo-
nia content in the air. The presence of normal atmospheric quantities of ammonia
does not accelerate zinc corrosion, and petrochemical plants where ammonium
salts are present show no accelerated attack on galvanized steel. However,
ammonia will react with atmospheric sulfur oxides, producing ammonium sul-
fate, which accelerates paint lm corrosion as well as zinc corrosion.
When ammonium reacts with NO
x
compounds in the atmosphere, ammo-
nium nitrite and nitrate are produced. Both compounds increase the rate of
zinc corrosion, but less so than SO
2
and SO
3
.
4.7.4.1 Zinc-Aluminum Hot Dip Coatings
Combinations of zinc and aluminum are the most important series of alloys.
These have been developed to improve the atmospheric corrosion resistance
of galvanized coatings. The two primary hot dip coatings are Galfan, which
is a zinc-5% aluminum alloy, and Galvalume, which is a zinc-55% alumi-
num-1.5% silicon alloy. Galfan coatings have a corrosion resistance up to
three times that of galvanized steel. The main difference between these two
coatings lies in the degree of cathodic protection they afford.
Galfan nds application in a mild urban-industrial atmosphere and in a
marine atmosphere. This latter application is particularly important because
the corrosion rate slows after about 4 yr, whereas conventional galvanized
steel shows rust in 5 yr. This slower rate of corrosion is evidence that the
zinc-5% aluminum coatings provide full cathodic protection to cut edges
over a longer period of time.
Galvalume provides good protection in urban-industrial atmospheres but
at the expense of reduced cathodic protection. The corrosion rate of these
coatings also tends to diminish after a year or two.
4.7.4.2 Zinc-Aluminum Thermally Sprayed Coatings
Zinc-15% aluminum coatings are available as thermally sprayed coatings.
These coatings have a two-phase structure consisting of a zinc-rich and
an aluminum-rich phase. The oxidation of the zinc-rich phase takes place
within the more inert aluminum-rich phase. The oxidation products formed
are encapsulated in the porous layer formed by the latter and do build up a
continuous surface layer as with pure zinc coatings. As a result, no thickness
112 Fundamentals of Corrosion
or weight loss is observed even after several years of exposure in atmospheric
eld testing.
4.7.4.3 Zinc-Iron Alloy Coatings
Compared with pure zinc coatings, the zinc-iron alloy coatings provide
increased corrosion resistance in acidic atmospheres but slightly reduced
corrosion resistance in alkaline atmospheres.
4.7.4.4 Cast Zinc Alloys
All the zinc-rich aluminum (ZA) alloys with an 8 to 29% aluminum con-
tent corrode less than 99.99% zinc. ZA-4 (zinc-4% aluminum) castings resist
atmospheric corrosion in much the same manner as pure zinc. One advan-
tage is that more zinc may be lost than with zinc coatings without affecting
the performance.
In general, corrosion resistance is improved by the addition of higher
amounts of aluminum. While ZA-8 (zinc-8% aluminum) and ZA-12 (zinc-
12% aluminum) exhibit only modest improvements over the standard 4%
alloy, ZA-27 (zinc-27% aluminum) with its signicantly larger amount of
aluminum-rich phase is much superior.
In plant atmospheres contaminated with either acidic or alkaline fumes, the
ZA alloys perform better than pure zinc; whereas in alkaline-contaminated
atmospheres, both zinc and the ZA alloys are more suitable than aluminum.
The addition of copper to zinc provides a slight improvement in corro-
sion resistance.
4.7. 5 Copper
For centuries, copper has been used as an architectural material. Copper
roofs are still in existence on many castles and monumental buildings that
are hundreds of years old.
When exposed to the atmosphere over long periods of time, copper forms
a coloration on the surface known as patina, which in reality is a corrosion
product that acts as a protective lm against further corrosion. The length
of time required to form the patina depends on the atmosphere because the
color is due to the formation of copper hydroxide compounds. Initially the
patina has a dark color, which gradually turns green. This is also known
as verdigras, which is French for green-gray. In urban or industrial atmo-
spheres, the compound is a mixture of copper/hydroxide/sulfate and in
marine atmospheres a mixture of copper/hydroxide/chloride. It takes
approximately 7 years for these compounds to form. When exposed to clean
or rural atmospheres, tens or hundreds of years might be required to form
the patina, or protective lm.

Atmospheric Corrosion 113
To be classied as copper, the compound must contain a minimum of 99.9%
copper. Elements such as silver, arsenic, antimony, phosphorus, tellurium,
nickel, cadmium, lead, sulfur, zirconium, magnesium, boron, and bismuth
may be present singularly or in combination.
Copper is noble to hydrogen in the electromotive force (emf) series and
thermodynamically stable with no tendency to erode in water and in non-
oxidizing acids free of oxygen. With copper and its alloys, the predominant
cathode reaction is the reduction of oxygen to form hydroxide ions. Therefore,
the presence of oxygen or other oxidizing agents is necessary for corrosion
to take place.
The coating of corrosion products forming the so-called patina has a com-
plex composition that varies from one location to another. Primary compo-
nents are generally copper oxide and one or more basic copper salts:
Basic sulfate: Cu (OH)
1:5
(SO
4
)
0.25
, Cu(OH)
1.33
(SO
4
)
0.33
Basic chloride: Cu(OH)
1.5
Cl
0.5
Basic carbonate: CuOH(CO
3
)
0.5
, Cu(OH)
1.33
(CO
3
)
0.33
, CuOH
0.67
(CO
3
)
0.6
Basic nitrate: Cu(OH)
1.5
(NO
3
)
0.5
Basic sulfate is predominant in urban atmospheres and basic chloride in
marine atmospheres, whereas in rural atmospheres the primary ingredient
is usually basic sulfate. At times, basic carbonate is found. The presence of
basic copper nitrate indicates that the water lm on the metal surface con-
tains appreciable amounts of nitrate.
In most cases, the rst products formed are Cu
2
O and CuO. These copper
oxides react slowly with pollutants in the air such as SO
x
, Cl
−
, CO
2
, and NO
2
forming the basic salts. For this reaction to proceed, the pH value of the sur-
face moisture must be sufciently high:
Cu O+0.5SO+1.5H O+0.75O2Cu(OH) (SO)
2222 1.540.
→
225
2221.5 0.5
Cu O+Cl +1.5HO+0.25O 2Cu(OH)Cl
−
→
Corrosion rates for copper in various atmospheres have been determined
as follows:
Atmosphere
Corrosion
Rate (µm/yr)
Rural 1
Urban and industrial 1–3
Marine 1–2

114 Fundamentals of Corrosion
The low corrosion rate and interesting patina formation of copper have been
responsible for its use in building structures such as roofs, facades, and gut-
ters. The blue-green patina forms after several years when conditions are
favorable in outdoor atmospheres. Initially it may take on a mottled appear-
ance but after 6 to 12 months, the surface usually turns a uniform dark brown
color and remains as such for several years. It can take 5 to 10 yr before a
green patina begins to appear on sloping surfaces in urban or marine atmo-
spheres. Because vertical surfaces have a much shorter time of wetness, they
remain black much longer. In a marine atmosphere, the surface facing the sea
acquires a green patina sooner than the other surface as a result of a greater
supply of chlorides from the sea winds.
In areas of very low pollution, the patina could conceivably take hundreds
of years to form because of the small supply of anions to form the copper salt.
When the temperature is very low or the exposure conditions are very dry,
the patina may not form at all.
Under acidic conditions, the green patina will not form. Such conditions
can occur near chimneys where acid smoke strikes the roof. Under these
conditions, the corrosion products are soluble.
Traces of dissolved copper will usually be found in rainwater running off
of copper-bearing surfaces. This takes place predominately in urban and
industrial areas that are highly polluted with SO
2
. Such rainwater can cause
blue staining on masonry, stonework, etc.
4.7.6 Copper alloys
To increase the strength and general usefulness of copper, it is alloyed with
other elements such as zinc, aluminum, nickel, tin, silicon, etc. The various
alloy families are as follows:
Principal Alloying Element Alloy Family
Zinc Brasses
Tin, aluminum, or silicon Bronzes
Nickel Copper nickels
There are other alloy families but these are of little importance in the eld of
corrosion resistance and will not be discussed.
4.7.6.1 Brasses
Brasses contain zinc as their principal alloying ingredient. Other alloying
ingredients include lead, tin, and aluminum. Lead is added to improve machi-
neability and not to improve corrosion resistance. The addition of approxi-
mately 1% tin increases the de-alloying resistance of the alloys. Aluminum is
Atmospheric Corrosion 115
added to stabilize the protective surface lm. Alloys containing in excess of
15% zinc are susceptible to de-alloying in acid or alkaline environments.
There is a type of corrosion in which brass dissolves as an alloy and the
copper constituent redeposits from solution onto the surface of the brass as
a metal in porous form. The zinc constituent may be deposited in place of an
insoluble compound or carried away from the brass as a soluble salt. The cor-
rosion can take place uniformly or locally. Uniform corrosion is more likely
to take place in acidic environments, while local corrosion is more apt to
take place in alkaline, neutral, or slightly acidic environments. The addition
of tin or arsenic will inhibit this form of corrosion. Conditions of the envi-
ronment that favor dezincication are high temperature, stagnant solutions
(especially of acid), and porous inorganic scale formation. Other factors that
stimulate the process are increasing zinc concentrations, and the presence of
both cuprous and chloride ions. As the de-alloying proceeds, a porous layer
of almost pure copper is left behind. This reaction layer is of poor mechani-
cal strength. The dezincication process on copper-zinc alloys is therefore
very detrimental.
High-zinc brasses are also susceptible to season cracking, which is a form
of stress cracking. The term originates from early in the twentieth century
when cartridge shells made of 70% copper and 30% zinc were found to
crack over a period of time. It was later realized that ammonia from decay-
ing organic matter in combination with residual stresses in the brass was
responsible for the cracking of these shells. This phenomenon was called
season cracking because the presence of high humidity during warm, moist
climates (or seasons) promoted the stress corrosion cracking.
Stress corrosion of brass commonly occurs when brass is subjected to an
applied or residual stress or while in contact with a trace of ammonia or
amine in the presence of moisture and oxygen. The risk of stress corrosion
cracking in brasses is greatest in industrial and urban atmospheres, charac-
terized by high contents of sulfur dioxide and ammonia. The stress corrosion
susceptibility is markedly lower in marine atmospheres. The relative resis-
tance to stress corrosion cracking of the brasses is as follows:
Low resistance:•
Brasses containing more than 15% zinc•
Brasses containing more than 15% zinc and small amounts of •
lead, tin, or aluminum
Intermediate resistance:•
Brasses containing less than 15% zinc•
The admiralty and the naval brasses are resistant to de-alloying as a
result of the addition of tin. High-zinc brasses resist suldes better than
low-zinc brasses.
116 Fundamentals of Corrosion
Alloys containing 15% or less zinc resist de-alloying and are generally more
resistant to stress corrosion cracking than the high-zinc alloys. Red brass is
a typical alloy of this group; it contains 15% zinc. Dissolved air, oxidizing
materials, and compounds that form copper complexes (e.g., ammonia) are
corrosive to the low-zinc brasses.
4.7.6.2 Bronzes
Copper-tin alloys are known as tin bronzes or phosphor bronzes. Phosphorus
is always present in small amounts even though tin is the principal alloying
ingredient. Phosphorus is used as an oxidizer and generally has a concentra-
tion of less than 0.5%
These alloys date back to the Bronze Age and probably are the oldest alloys
known. Many of the artifacts produced during the Bronze Age are still in
existence. Items such as bells, statues, vases, and swords have survived hun-
dreds of years of exposure to a wide variety of environments, proving the
corrosion resistance of these materials.
Copper-aluminum alloys are known as aluminum bronzes. They are com-
plex alloys containing iron, nickel, silicon, manganese, and other elements
as well as aluminum.
As with copper, the aluminum bronzes nd many architectural applica-
tions. A special aluminum bronze alloy designated C-61500 has been speci-
cally developed for architectural use. It is reported to have a lm resistance
20 times that of copper C-11000. These are suitable for marine atmospheres.
4.7.6.3 Copper-Nickel Alloys
The copper-nickel alloys are referred to as cupronickels and have nickel as
the principal alloying ingredient. Of all the copper alloys, the cupronickels
are the most resistant to stress corrosion cracking in ammonia and ammo-
niacal environments. These alloys do not nd many applications to resist
atmospheric corrosion because of their cost. They would be most suitable for
marine atmospheres.
4.7.7 aluminum alloys
The resistance to atmospheric corrosion of aluminum is due to the rapid for-
mation of a thin, compact, adherent oxide lm over the surface that limits cor-
rosion. This surface lm, when formed in air, is approximately 10 to 20 mm
thick. It is thicker when formed in the presence of water or water vapor. This
oxide lm is stable (insoluble) in the pH range of approximately 4 to 9, which
includes many atmospheric environments.
Pourbaix potential-pH diagrams can be used to predict the regions of
oxide stability and of uniform corrosion in atmospheric types, as shown in
Figure 4.3. Note that aluminum is thermodynamically stable only at low
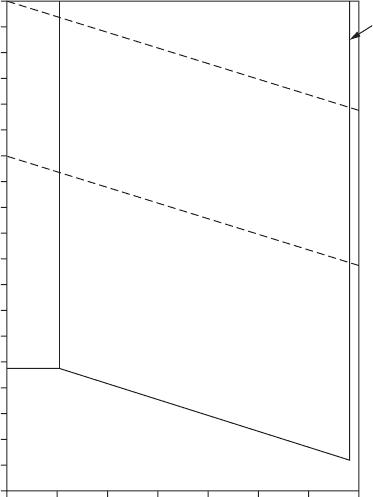
Atmospheric Corrosion 117
potentials. Because of its property of developing a protective coating of alu-
mina, it can be used in the presence of water. At an Al
3+
activity of 10
−1
µ, the
stability range of the oxide extends down to pH 2.5.
Corrosion products that form during the outdoor corrosion of aluminum
are amorphous Al(OH)
3
, or ∝-Al(OH)
3
(bayerite), and γ-Al
2
O
3
, the latter with
varying amounts of water in the lattice.
A 10- to 20-mm thick amorphous oxide protective coating forms when a
fresh aluminum surface is exposed to dry air. In a moist atmosphere, the
oxide coating will thicken. The coating consists of one dense, protective bar-
rier layer next to the metal and an outer, more permeable bulk layer.
Water-soluble salts such as Al
2
(SO
4
)
3
may be formed when anions (e.g.,
SO
4
2−
and Cl
−
) are deposited on the oxide surface and react with the oxide
lm. They may also be incorporated into the lattice to form a variety of basic
salts and complexes.
In urban atmospheres, the oxide coating is protective against SO
2
pollu-
tion, producing a relatively low pH value in the moisture lm. If high SO
2
pollution exists, a very low pH value of the moisture lm is produced, which
leads to the dissolution of the protective lm.
14121086420
–2.6
–2.4
–2.2
–2.0
–1.8
–1.6
–1.4
–1.2
–1.0
–0.8
–0.6
–0.4
–0.2
0
0.2
0.4
0.6
Al
3+
Al
2
O
3
3H
2
O
AlO
2
–
Al
Potential, V (SHE)
pH
0.8
1.0
1.2
FigurE 4.3
Potential-pH diagram, Al-H
2
O; 25°C, 10
−1
MAl. (Source: From Reference 6.)
118 Fundamentals of Corrosion
In the presence of chloride ions, pitting may be initiated. It is believed
that the chloride ions migrate to the oxide layer and lower its resistance to
outward migration of Al
3+
. During the propagation stage, aluminum is dis-
solved anodically to Al
3+
ions within the pit. A cathodic reaction takes place
either inside the pit, close to the mouth, or outside the pit and consists of the
reduction of oxygen or Η
+
ions. The passivating oxide layer has low elec-
tronic conductivity but the cathode reaction may locally destroy the protec-
tive oxide layer due to alkalization. This lowers the electrode potential and
may even make hydrogen liberation possible. Acid conditions are created
within the pit by hydrolysis of the Al
3+
ions, and a cap of Al(OH)
3
and/or
Al
2
O
3
forms over the mouth of the pit. The corrosion products nally block
the operation of the pit.
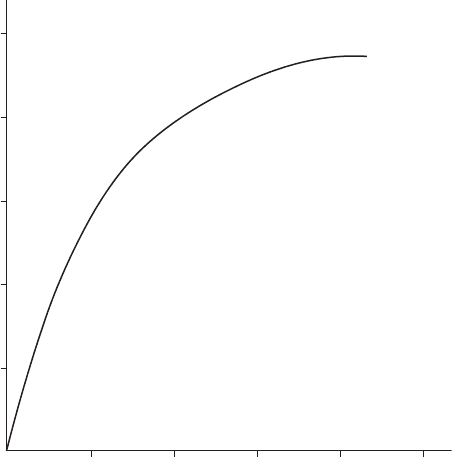
Aluminum retains its shiny appearance for years in clean outdoor atmo-
spheres. Small pits, which are barely visible to the naked eye, develop in
polluted outdoor atmospheres. These pits become covered with crusts of
aluminum oxide and aluminum hydroxide. During the rst few years of
exposure, the growth rate of the maximum pit depth is relatively high. This
growth rate decreases gradually, so that the pit depth approaches a nearly
constant value (see Figure 4.4).
1086
Exposure Time, y
42
20
40
Maximum Pit Depth, µm
60
80
100
FigurE 4.4
Maximum pit depth vs. exposure time for Al Mn 1.2 in an urban atmosphere.
